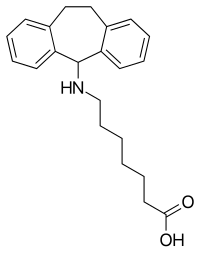
Amineptine
 | |
| Clinical data | |
|---|---|
| Trade names | Survector, others |
| Synonyms | S-1694 |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Elimination half-life | Amineptine: 0.8–1.0 hours[1][2] Metabolite: 1.5–2.5 hours[1][2] |
| Excretion | Renal |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.055.271 |
| Chemical and physical data | |
| Formula | C22H28NO2 |
| Molar mass | 338.4653 g/mol |
| 3D model (JSmol) | |
| (verify) | |
Amineptine was developed by the French Society of Medical research in the 1960s.[5] Introduced in France in 1978 by the pharmaceutical company Servier,[6] amineptine soon gained a reputation for abuse due to its short-lived, but pleasant, stimulant effect experienced by some patients. (This is to be distinguished from its antidepressant effect, which appears in approximately seven days after commencing treatment.)
After its release into the European market, cases of hepatotoxicity emerged, some serious. This, along with the potential for abuse, led to the suspension of the French marketing authorization for Survector in 1999.[7]
Amineptine was never approved by the U.S. Food and Drug Administration (FDA) for marketing in the United States, meaning that it is not legal to market or sell amineptine for any medical uses in the U.S.
Contents
[hide]After Note: To me it is for major depressive period before it gets too late and needs E.C.T because it maybe habit forming . I cannot know it is true or not as I am not a Doctor.

